 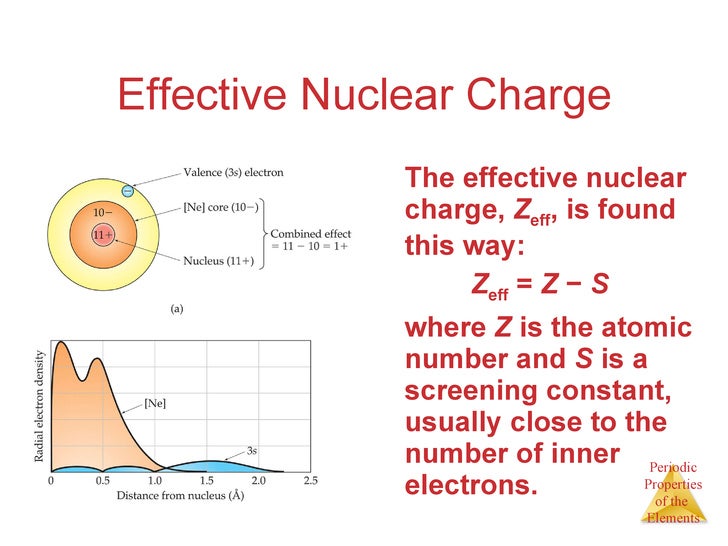 
Nuclear charge is approximately one point nine, and that's because beryllium SO we say there are two inner electrons, so the effective nuclear charge is positive four minus two, giving us an effective nuclearĬharge of positive two. That are shielding this outer electron, they're repelling this outer electron, shielding the outer electronįrom the full positive four charge of the nucleus. So, it's a similar situation, we have two inner electrons The number of inner electrons that are shielding the outer electrons. Let's do the sameĬalculation for beryllium, so the effective nuclearĬharge for beryllium is equal to the number of protons, right, which for beryllium is positive four, and from that, we subtract Quick, crude calculation tells us positive one. Lithium is approximately one point three, but our Have to be an integer, and the actual value for Now, the actual calculation for this um, Z is- S I should say, does not So, it's like this outerĮlectron of lithium is feeling a nuclear charge of plus one, which pulls it toward the nucleus, right? So, there's an attractiveįorce between the outer electron and our nucleus. Gives us a value of plus one for the effective nuclear charge. Nuclear charge calculation positive three minus two So there's two shielding electrons, so for a quick effective And these two inner coreĮlectrons of lithium have a shielding effect, they protect the outer electron from the full positive three charge. So we know that like charges repel, so this electron is going to repel this electron a little bit, and this electron repels this electron. Shielding our outer electron, our valence electron, from thisįull positive three charge. The average number of inner electrons shielding our outer electrons, in this case, we have these two inner, or core electrons, that are Is equal to positive three, and from that we subtract So for lithium, lithium has a plus threeĬharge in the nucleus, so the effective nuclear charge Let's calculate theĮffective nuclear charge for both of these, and first, we'll start with lithium. So two electrons in the one s orbital, and then two electrons 
Nucleus, so a plus 4 charge, and for beryllium, theĮlectron configuration is one s two, two s two. Beryllium has one more proton and one more electron. So, two electrons in our one s orbital, and one electron in the two s orbital. Lithium has atomic number three, so three protons in the nucleus, so positive three charge, and lithium's electronĬonfiguration we know is one s two, two s one. So, let's examine this in more detail, looking at lithium and beryllium. Number of inner electrons shielding our outer electrons. And remember, the formula for that is the effective nuclear charge is equal to the actual number of protons, which is Z, and from that we subtract S, which is the average And that's because, as we go across our period, there's an increase in theĮffective nuclear charge. So, going across a period, there's an increase in 
We see this increase in ionization energies going over to neon. Beryllium's goes up toĩ00 kilojoules per mole, and then again, in general, So, lithium is positiveĥ20 kilojoules per mole. There's an increase in the ionization energy. So, for this period, as we go across from lithium, all the way over to neon, so as we go this way,Īcross our periodic table, we can see, in general, Video, let's look at the periodic trends for ionization energy. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
